Aeration constrain is bioreactors
Dissolved oxygen is important during cultivation. Since oxygen is sparingly soluble in water, it may be the growth-limiting factor in these bioreactors. The solubility of oxygen in culture media is ~6,6 µg/mL at 37°C when exposed for a 5 % CO2 / p5% air gas mixture. The typical Oxygen Uptake Rate for mammalian cells are between 3x10-10 and 2x10-8 mg/cell/hour. For optimum growth it is therefore important to maintain the dO2 above this critical level by aeration. Of course, to be effective, the mass transfer rate from the gas bubbles to the liquid broth must equal or exceed the rate at which growing cells Oxygen Uptake Rate. In general terms the air or mixed gas volume required is ranging one litre/working volume/hour.
- The classical aeration method for bioreactors is performed by sparging (bubbling) gas through the bioreactor media. The gas being air or mixed gases by slight over-pressure forced through small holes or pores in the sparger tip. The smaller the bubbles the larger surface area and hereby improved oxygen diffusion through the bubble to liquid contact.
- Surface aeration is obtained through the media surface through the head space. And sufficient for limited cell density culture in small vessels, such as T-flasks, roller bottles, etc.
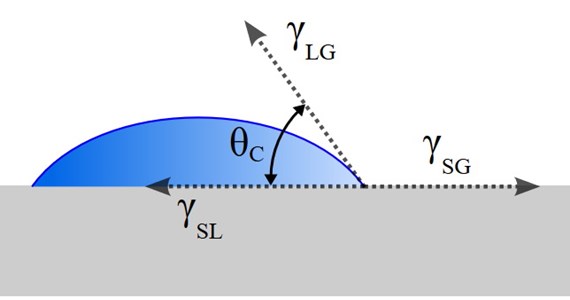
- Contact angle is the interaction between a liquid drop and a solid surface. Measured in degrees (0-180°) where most polymers have contact angles above 90° and many metals and glass have less than 90° contact angle. Tested by adding a drop of water. Very important to observe and realise contact angles when a gas pass a porous body made of solids for aeration purposes in a bioreactor. Wettability also expressed as hydrophobic or hydrophilic solid surfaces.
- Membrane aeration replaces the need for gas sparging by providing a large interfacial area for oxygen diffusion. Membrane aeration is able to avoid contact between cells and bubbles. Not a method used for re-usable bioreactors as to maintenance issues. Though highly relevant for Single-Use-Bioreactors and will be pursued by CerCell.
Oxygen transfer via sparging is usually limited by the liquid film surrounding the gas bubbles. The rate of transport is given by:
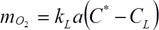
where kL is the oxygen transport coefficient (cm/h), a is the gas-liquid interfacial area (cm2/cm3), kLa the volumetric oxygen transfer coefficient (h-1), C* is saturated dO2 concentration (mg/l) (approx. 8 mg/l at 25 deg. C and 1 atm.), CL is the actual dO2 concentration in the liquid (mg/l), and NO2 is the rate of oxygen transfer (mg O2/ l/h).
More info under Download/Science work and the page Aeration Available.